NeuroAD, a novel medical device for Alzheimer’s treatment, has shown promising results in clinical trials. The lack of side-effects, the cognitive improvements in patients and the modification of the disease’s course indicate that this non-invasive therapy may bring significant cognitive improvement to AD patients.
“A serious disease of the cerebral cortex”
In 1906, at the 37th Meeting of South-West German Psychiatrists, Dr. Alois exposes the singular case of Auguste D. Back then, Auguste’s illness was identified as a “characteristic serious disease of the cerebral cortex”; today, it is known that Auguste was the first reported case of Alzheimer’s disease (AD) [1].
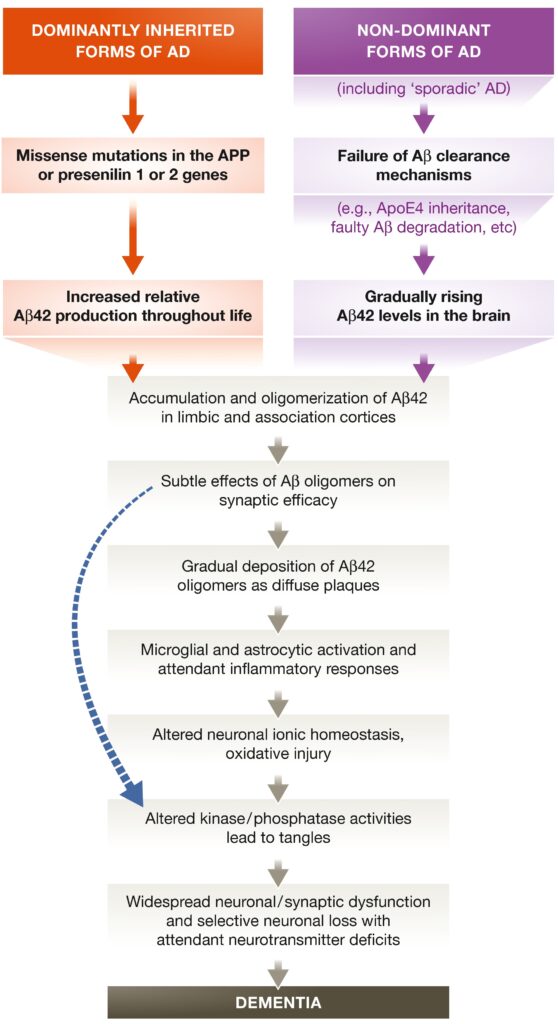
At the age of 51, Auguste was admitted in 1901 at a psychiatric clinic in Frankfurt, Germany, with symptoms of hallucinations, delusions and cognitive impairment. There, she was examined and carefully followed by Dr. Alois Alzheimer. After a severe aggravation of Auguste’s condition and posterior death, Dr. Alzheimer examined her brain and found the typical hallmarks of AD: “abnormal clumps” and “tangled bundles of fibers”. Further investigations determined that those clumps are actually deposits of β-amyloid plaques. In addition, the examination of Auguste’s brain confirmed that this was not a usual vascular dementia, but rather the case of a degenerative disease [2].
Origin and symptoms
One hundred and ten years later, several theories have been put forward to explain the trigger for the initiation of AD. Although one of the most contested, the Aβ hypothesis is perhaps the most currently used. The abnormal deposition of β amyloid proteins in the brain causes a disruption of the synaptic activity and the aberrant functioning of the neurotransmitters [3]. The accumulation of the subtle changes in the synaptic and neuronal activity in the brain eventually leads to a more severe form of dementia.
In the mild phase of the disease, which lasts from 2 to 4 years, AD patients often experience a drop in energy levels, loss of recent memory, mood swings and mild coordination problems. As the disease progresses, patients enter the most severe phase, which usually lasts 1 to 3 years. In this stage, people cannot express themselves, remember things, process information and they can even experience hallucinations or seizures.
Therapies
No drug has yet been proven to be effective in achieving a cure to AD. Thus, most of the medication in the market aims at treating or temporarily alleviating the symptoms of the disease.
However, the next-generation medicine is slowly permeating the market. Those new medicines aim to modify the disease progression itself. Some of the most promising examples currently in investigation target the β-amyloid protein, by blocking the enzyme that produces the protein or by preventing the clumping of Aβ in the brain. Other drugs target enzymes involved in the clumping and formation of tangles in the brain, one the hallmarks of the diseases. Some of those targets include the beta-secretase enzyme, the Tau protein or the 5HT6 receptor.
NeuroAD
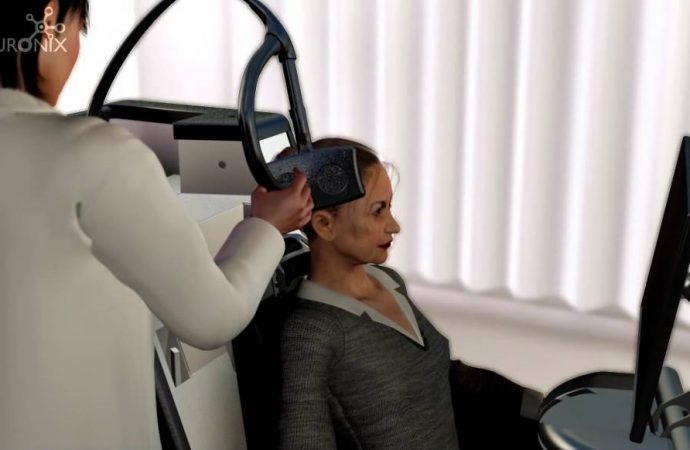
Recently, a novel AD treatment has entered the European market and after completing clinical trials in the U.S., is currently under FDA review to market in the U.S. Safely used in conjunction with all medications, the NeuroAD technology utilizes repetitive Transcranial Magnetic Stimulation (rTMS) with Cognitive training. rTMS is a non-invasive method of brain stimulation, that has been widely used for the treatment of depression and other neurological disorders, with minimal side effects [4]. Although the patterns and repetitions of rTMS can vary between different therapies, significant benefits in the brain plasticity have been reported in almost every study of TMS.
During the intensive 6-week treatment, patients receive daily sessions of TMS while, at the same time, receive tailored exercises that meet their cognitive needs. The combination of TMS and Cognitive Training has been shown to have a synergistic effect and induce Long-Term Potentiation (LTP). LTP is the stable, long lasting increase in synaptic activity that drives the processes of learning and memory formation.
Cognitive improvements
Clinical data showed that patients who received the NeuroAD treatment increased their levels of LTP. According to the experts at Neuronix Medical, “patients start to feel improvement in cognitive ability as early as three to four weeks after the treatment begins”. Throughout the course of the treatment, these improvements should increase , and have also been documented to have a long-term impact of up to a year.
The realization of Positron Emission Tomography (PET) scans is underway in some of the institutions that own the necessary equipment to perform it. This technique will allow physicians to investigate the presence of Aβ deposits in the brain, one of the most predominant hallmarks of the disease.
References:
[1]Maurer, K. et al (1997). Auguste D and Alzheimer’s disease The Lancet, 349 (9064), 1546-1549 DOI: 10.1016/S0140-6736(96)10203-8
[2] Jellinger, K. (2006). Alzheimer 100 – highlights in the history of Alzheimer research Journal of Neural Transmission, 113 (11), 1603-1623 DOI: 10.1007/s00702-006-0578-3
[3] Selkoe, D., & Hardy, J. (2016). The amyloid hypothesis of Alzheimer’s disease at 25 years EMBO Molecular Medicine, 8 (6), 595-608 DOI: 10.15252/emmm.201606210
[4] Gersner, R., Kravetz, E., Feil, J., Pell, G., & Zangen, A. (2011). Long-Term Effects of Repetitive Transcranial Magnetic Stimulation on Markers for Neuroplasticity: Differential Outcomes in Anesthetized and Awake Animals Journal of Neuroscience, 31 (20), 7521-7526 DOI: 10.1523/JNEUROSCI.6751-10.2011
Sources:
Neuronix Medical – procedure
Neuronix – Clinical data
Image Credit: Gal Edri