Hydrolates or flower waters are known for their antimicrobial properties and are widely used in food, pharmaceutical, and cosmetological industries. Rose water, orange blossom water, and lavender water are probably the most popular ones. Hydrolates can be prepared from any kind of flower, but their properties vary, some of them may cause allergic reactions. The best example is the limonene from lemon/orange. In this article, we will concentrate on flower waters that have shown beneficial properties.
Benefits of using hydrolates
Hydrolates have softer action than essential oils, making them an excellent base for the production of soaps, lotions, and creams. For example, the active constituent of carnation water is eugenol—a chemical compound known for its antiseptic properties. Geranial water, which contains geraniol, is appreciated for antifungal and antiseptic properties. Lavender water is used as a soothing, healing, and antibacterial cosmetic because it consists of valuable compounds such as rosmarinic, ursolic, and carnosic acid.
What are hydrolates made of?
Hydrolates have been trendy recently due to their natural, organic composition. When you look at their label, it reveals a short list of essential oils components. This is because hydrolates are obtained by hydrodistillation of different parts of a plant, so the product mainly consists of volatile ingredients in essential oils and the water used for distillation. Hydrodistillation is a process used to extract components that only involve the plant and water.
Hydrolates are used mostly for their natural scent, so one might ask: why not use essential oils instead? After all, essential oils and hydrolates share a large part of their composition – the main difference is that they are more diluted than essential oils.
Well, while essential oils have a powerful scent, the hydrolates are somewhat delicate, so it is a matter of preference or purpose which one to choose. If a powerful scent is desired, such as a perfume, one should use essential oils. On the other hand, if you need delicacy—that’s when hydrolates may come in handy.

Let’s make the magic happen: hydrolates preparation
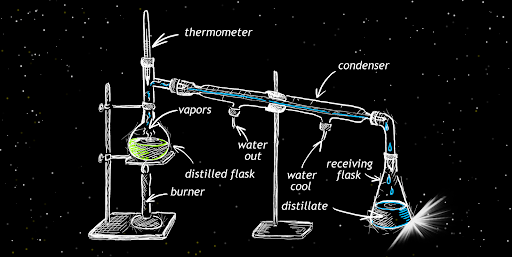
Generally speaking, steam distillation, or simply hydrodistillation, is a process in which compounds that don’t dissolve in water undergo isolation from a mixture. The preparation is as follows: first, heat water in a flask at the boiling point to create steam. Then, transport the steam to a flask containing a plant compound that is also under heat; see image below. Under these conditions, the essential oils undergo a phase transition from liquid to a gaseous state.
Finally, transport the gaseous essential oils-water mixture to a condenser, where it will revert to a liquid state. Then, use gravity to transport the liquid mixture to a flask. This will create a beautifully smelling hydrolate.
While regular distillation is simpler than steam distillation, the latter is carried out in far lower temperatures so that compounds don’t undergo thermal decomposition. According to the 300 years old John Dalton’s law, the sum of partial pressures of individual gases is equal to the total pressure. Because boiling occurs when the system reaches total pressure equal to the atmospheric pressure, far less heat is needed to make essential oils boil if you first introduce water as steam. Thus, a hydrolate can be obtained without decomposing its ingredients.
Rationalizing the magic: chemistry behind hydrolates
Hydrolates composition may vary depending on what plant and which part of it was used for hydrodistillation. Floral waters contain many different organic molecules but mainly consist of compounds called terpenes, which gives Hydrolates their amazing aroma. Terpenes are a large family of substances composed of subunits with five carbon and eight hydrogen atoms arranged in different ways. Almost all terpenes can be only obtained from living organisms and are challenging to synthesize in the laboratory.
Terpenes determine the smell of some parts of plants that we encounter daily. For example, the scent of lemons is determined by a terpene called limonene. On the other hand, pinene gives pines their beautiful Christmas aroma, and geraniol accounts for the smell of geranium and grapes. They also display antifungal and antiseptic properties. For example, a compound named taxol and its derivatives is widely used as drugs in chemotherapy.
Because terpenes are volatile, they slowly evaporate from the hydrolate, diffusing near our noses and adsorbed on the smell receptors. Then, the electrical signal is transported to the temporal lobe in our brain, where it is interpreted as a rather lovely scent.
Although hydrolates are less known than essential oils, they are just as effective. They are delicate and less active than essential oils and are perfect for people with allergies, sensitive skin, and babies. Another use of them is to help with emotional instability and lousy mood. Essential oils are known for calming and relaxing properties. Applying these natural wonders in everyday life is beneficial for our health, mood, and well-being.
This article is a joint work of Adrian Romaniuk (Faculty of Chemistry, University of Warsaw), Patrycja Hejduk (Faculty of Chemistry, University of Warsaw), Agnieszka Pregowska (Institute of Fundamental Technological Research, Polish Academy of Sciences), and Magdalena Osial (Faculty of Chemistry, University of Warsaw). Image Credit: Magdalena Osial.
References
[1] Šilha, D.; Švarcová, K.; Bajer, T.; Královec, K.; Tesařová, E.; Moučková, K.; Pejchalová, M.; Bajerová, P. (2020). Chemical Composition of Natural Hydrolates and Their Antimicrobial Activity on Arcobacter-Like Cells in Comparison with Other Microorganisms. Molecules 25(5654). DOI: 10.3390/molecules25235654
[2] Acimovic, M; Tešević, V.; Smiljanic, K.; Smiljanic, K.; Sikora, V. (2020). Hydrolates: By-products of essential oil distillation: Chemical composition, biological activity, and potential uses. Advanced Technologies 9(2):54-70. DOI: 10.5937/savteh2002054A
[3] Prusinowska, R.; Śmigielski, K.; Stobiecka, A.; Kunicka-Styczyńska, A. (2016). Hydrolates from lavender (Lavandula angustifolia) – their chemical composition as well as aromatic, antimicrobial, and antioxidant properties. Natural Product Research 30(4), 386-93. DOI: 10.1080/14786419.2015.1016939
[4] Yuwono, M.; Siswandono; Hafid, A. F.; Poernomo, A. T.; Agil, M.; Indrayanto, G.; Ebel, S. (2002). Eugenol. Analytical Profiles of Drug Substances and Excipients 9, 149-177. DOI: 10.1016/S1075-6280(02)29006-0
[5] Aldred, E. M.; Buck, C.; Vall, K. Chapter 22 – Terpenes. Editor(s): Aldred, E. M.; Buck, C.; Vall, K. Pharmacology 2009, 167-174. Churchill Livingstone. DOI: 10.1016/B978-0-443-06898-0.00022-0.
[6] Buckle, J. (2015). Chapter 3 – Basic Plant Taxonomy, Basic Essential Oil Chemistry, Extraction, Biosynthesis, and Analysis. Clinical Aromatherapy 37-72.
[7] Kupska, M; Gromadzka, J.; Namieśnik, J. Terpenes: Properties and determination. International Conference on Food Safety and Regulatory Measures 2015, August 17-19, Birmingham, UK.
[8] Joshee, N.; Dhekney, S. A.; Parajuli, P.; Agricultural Research Station, Fort Valley State University, Fort Valley, GA USA Department of Agriculture, Food and Resource Sciences, University of Maryland Eastern Shore, Princess Anne, MD USA. Therapeutic and Medicinal Uses of Terpenes 2019.
[9] Genet, T. Hydrolates or floral water: a soothing treatment for your skin, online: http://www.magicbeansandhappiness.com/?p=10813&lang=en (available: June 28, 2021).
[10] Saiyudthong, S.; Marsden, C. A. (2011). Acute effects of bergamot oil on anxiety-related behavior and corticosterone level in rats. Phytotherapy Research 25(6),858-62. DOI: 10.1002/ptr.3325






[…] post Hydrolates: natural solutions as effective as essential oils first appeared on United Academics […]